 Note: At the point when high voltage is applied, it ionizes the gas and it is positive ions of gas that establishes the canal ray. Hence, we can say that option A is the correct option. A portion of the positively charged particles convey multiples of the fundamental value of the charge. The conduct of particles in an electric and magnetic field was inverse to that of cathode rays.Ĥ. The charge to mass proportion for the particles of rays was seen as various for various gases.ģ. It is on the grounds that the canal rays are made out of positive ionized particles framed by ionization of gas present in the tube.Ģ. Not at all like cathode rays, canal rays rely on the nature of gas present in the tube. These rays were moving the other way of cathode rays and were named canal rays.ġ. At the point when the voltage was expanded to a few thousand volts, a faint luminous ray was seen reaching out from the holes in the back of the cathode.ĥ. The electricity began streaming as the circuit was finished.Ĥ. High voltage is passed to the two metal pieces in order to ionize the air and make it a channel of electricity.ģ. Apparatus was set up by giving a high voltage source and clearing the air to keep up low pressure inside the tube.Ģ. A swoon luminous ray was seen reaching out from the holes in the rear of the cathode.ġ. In the experiment, Goldstein applied high voltage over a discharge tube which had a perforated cathode. The disclosure of protons which occurred after the revelation of electrons further fortified the structure of the atom. The Canal Ray experiment is the experiment performed by German researcher Eugen Goldstein in 1886 that prompted the revelation of the proton. 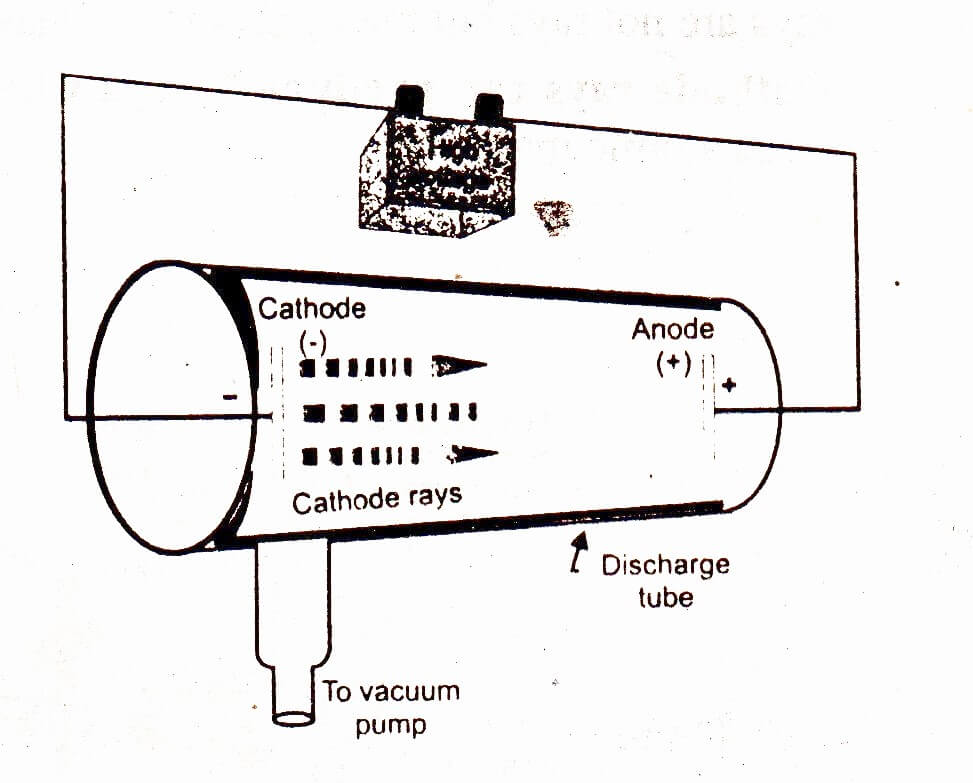
Canal rays originate in the region among cathode and anode in a discharge tube. 
Protons are positively charged particles. Hint- Canal rays are likewise called anode rays or positive rays. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
